Internal conversion of polyenes
1,3,5-cycloheptatriene (CHT) can be used as a model system of polyenes, which form a basic group of molecules in photo- and biochemistry. Prominent examples are rhodopsin in the process of vision or the vitamin D system. The photochemically allowed [1,7]-sigmatropic hydrogen migration (H shift) in CHT is a classical example of a pericyclic reaction associated with internal conversion processes. For this type of reaction, it is believed that a symmetric dark state is involved as has been demonstrated for the cyclization of butadiene. As a consequence the internal conversion is supposed to proceed in two stages as sketched below:
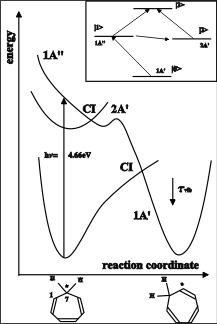
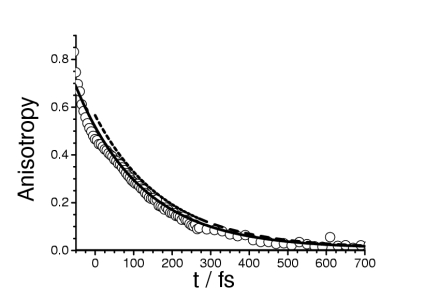
This process has been studied in 1,3,5-cycloheptatriene (CHT) and its perdeuterated counterpart (CHT-d8) in liquid cyclohexane by femtosecond pump-probe experiments with 50 fs time resolution. Temporal absorption and transient anisotropy profiles were monitored. The experimental findings are consistent with the following mechanism: After excitation, the system undergoes a fast internal conversion associated with a molecular reorientation. The characteristic times are (110 ± 10) fs for CHT and (150 ± 20) fs for CHT-d8. If the relaxation process of an excited state is accompanied by any orientational change of the transition dipole moment, transient anisotropy spectroscopy provides information on the temporal evolution of the molecular orientation. As the relaxation time constants both of the transient absorption profiles and the transient anisotropy are comparable, we attribute this transient change for CHT its perdeuterated counterpart CHT-d8 to the photochemically induced [1,7]- sigmatropic H shift.
Further reading:
[1] A. Hertwig, H. Hippler, H. Schmid, and A.-N. Unterreiner, “Direct time-resolved UV-absorption study on the Ultrafast Internal Conversion of Cycloheptatriene in Solution”
Phys. Chem. Chem. Phys., 1, 5129-5132 (1999)
[2] A. Hertwig, H. Hippler and A.-N. Unterreiner, “Reply to the Comment on ‘Direct time-resolved UV-absorption study on the Ultrafast Internal Conversion of Cycloheptatriene in Solution’”
Phys. Chem. Chem. Phys., 2, 1437-1438 (2000)
[3] H. Hippler, M. Olzmann, O. Schalk, and A.-N. Unterreiner, “Pump-probe spectroscopy of cycloheptatriene: Transient anisotropy and isotope effect”
Z. Phys. Chem., 219, 389-398 (2005)
